Aseptic Processing – Essential Personnel Practices
Coming Soon!
Personnel play a critical role in ensuring product sterility and maintaining compliance in aseptic manufacturing environments. An operator’s movements, behavior, and gowning practices directly impact product integrity and, ultimately, patient safety.
Through an interactive 3D aseptic processing environment, Skillpad’s Essential Personnel Practices e-Lesson provides the critical ‘how’ and ‘why’ insights needed for effective contamination control. Users gain essential knowledge in gowning procedures, cleanroom hygiene, aseptic behavior, material handling, and contamination prevention – all aligned with EU Annex 1 requirements.
Description
- Regulatory Compliance with EU Annex 1: Understand key personnel-related requirements for aseptic processing and how to align daily cleanroom practices with regulatory expectations.
- Proficiency in Aseptic Gowning: Learn step-by-step gowning procedures for different cleanroom grades (Grade B, Grade C) to prevent contamination and maintain controlled environments.
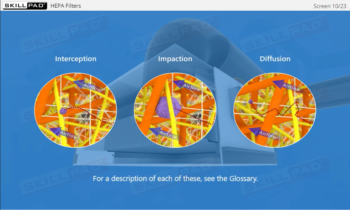
- Enhanced Awareness of Contamination Control: Recognize the impact of personnel behaviors, movement, and hygiene on minimizing contamination risks and protecting product sterility.
- Application of Best Practices in Aseptic Environments: Engage in interactive scenarios that reinforce proper cleanroom conduct, aseptic handling, and contamination prevention strategies.
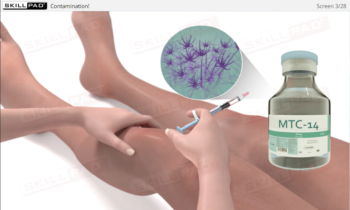
- Confidence in Addressing Non-Compliance: Develop the ability to identify, respond to, and correct personnel-related deviations that could compromise aseptic integrity.
Learning Objectives
- Identify key EU Annex 1 regulatory requirements for personnel working in aseptic processing of sterile medicinal products.
- Describe the correct gowning procedures for different cleanroom grades to minimize contamination risks.
- Recognize critical aseptic behaviors that support contamination control and protect product sterility in cleanrooms.
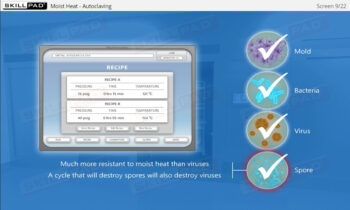
- Explain essential health and hygiene standards required to prevent contamination risks in aseptic environments.
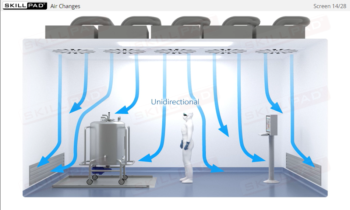
- Apply best practices for personnel movement and conduct to reduce particle shedding and maintain controlled conditions.
- Demonstrate an understanding of common personnel-related non-compliances and the corrective actions needed to uphold aseptic integrity.
Keywords
- Aseptic Processing
- Cleanroom Behavior
- Gowning Procedures
- Contamination Prevention
- EU Annex 1 Compliance
- Personnel Practices
- Sterile Manufacturing
- Aseptic Technique
Module Features
Animations
Voice Over
Knowledge Checks
Assessments
SCORM/AICC compatible
Full Screen