Aseptic Processing – Working with Isolators
Coming Soon!
A detailed look at the use of closed isolators in aseptic processing of sterile products, covering their design, operating principles, and critical role in contamination control. This module explores how isolators maintain a sterile environment using HEPA filtration, positive pressure, and controlled airflow. Learners will gain practical knowledge of essential procedures, including preparation, aseptic manipulations, material transfers, and decontamination, ensuring proper use and regulatory compliance. Engaging interactive elements, including 3D simulations and scenario-based learning, reinforce best practices for working within an isolator.
Description
- Comprehensive Knowledge of Closed Isolators
Gain a deep understanding of closed isolators, their components, and their function in aseptic processing to maintain sterility and ensure product integrity.
- Critical Insights into Contamination Control
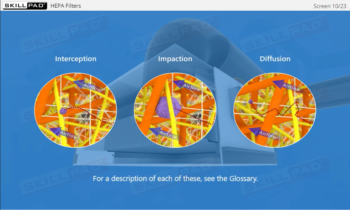
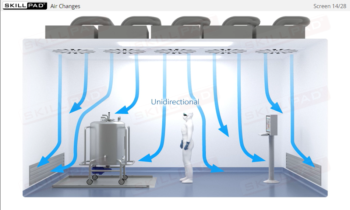
Learn how HEPA filtration, unidirectional airflow, and positive pressure work together to prevent microbial contamination, ensuring compliance with stringent regulatory standards.
- Proficiency in Aseptic Technique and Best Practices
Develop essential skills for working within an isolator, including proper glove use, material transfers, and aseptic manipulations—all reinforced through interactive 3D simulations and real-world scenarios.
- Confidence in Preparation and Decontamination Procedures
Understand the critical steps for preparing an isolator, from verifying equipment status to implementing personal protective measures. Learn decontamination procedures that maintain sterility for successive operations, reducing contamination risks.
- Interactive and Applied Learning
Engage with hands-on learning experiences, including guided discovery activities, real-time decision-making exercises, and knowledge checks to reinforce key concepts and best practices.
Learning Objectives
- Explain the purpose and function of a closed isolator in aseptic processing.
- Describe the key applications of closed isolators in the pharmaceutical and biotechnology industries.
- Identify the major components of a closed isolator and their roles in maintaining sterility.
- Explain the operating principles of a closed isolator.
- Outline the preparation steps for working in a closed isolator, including equipment verification and use of Personal Protective Equipment (PPE).
- Describe best practices for aseptic technique within a closed isolator, including glove use, material transfers, and contamination prevention.
- Apply best practices for working in a closed isolator.
- Explain the decontamination procedures for a closed isolator.
Keywords
- Aseptic Processing
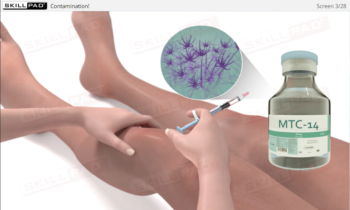
- Contamination Prevention
- Closed Isolator
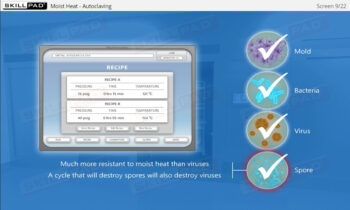
- Decontamination
- Glove Ports
- Material Transfer
- Sterile Products
- Best Practices
Module Features
Animations
Voice Over
Knowledge Checks
Assessments
SCORM/AICC compatible
Full Screen